Hello everyone
I'm here to share my views on ozone layer depletion.
Ozone, or trioxygen, is an inorganic molecule with the chemical formula O ₃. It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope O ₂, breaking down in the lower atmosphere to O ₂.
I'm here to share my views on ozone layer depletion.
What is Ozone layer?
Ozone, or trioxygen, is an inorganic molecule with the chemical formula O ₃. It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope O ₂, breaking down in the lower atmosphere to O ₂.Ozone depletion
Image of the largest Antarctic ozone hole ever recorded over the South Pole in September 2006
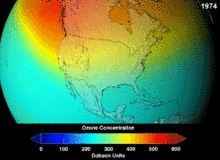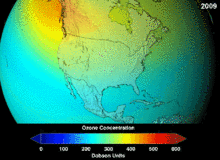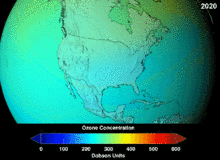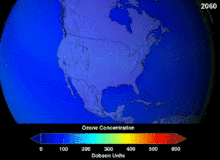
NASA projections of stratospheric ozone concentrations if chlorofluorocarbons had not been banned
Ozone depletion consists of two related events observed since the late 1970s: a steady lowering of about four percent in the total amount of ozone in Earth's atmosphere (the ozone layer), and a much larger springtime decrease in stratospheric ozone around Earth's polar regions.The latter phenomenon is referred to as the ozone hole. There are also springtime polar tropospheric ozone depletion events in addition to these stratospheric events.
In 2019, NASA announced the "ozone hole" was the smallest ever since it was first discovered in 1982.
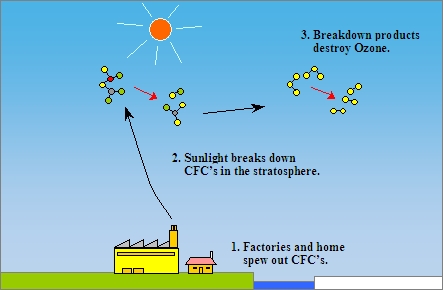
The main cause of ozone depletion and the ozone hole is manufactured chemicals, especially manufactured halocarbon refrigerants, solvents, propellants and foam-blowing agents (chlorofluorocarbons (CFCs), HCFCs, halons), referred to as ozone-depleting substances (ODS). These compounds are transported into the stratosphere by turbulent mixing after being emitted from the surface, mixing much faster than the molecules can settle.Once in the stratosphere, they release halogen atoms through photodissociation, which catalyze the breakdown of ozone (O3) into oxygen (O2). Both types of ozone depletion were observed to increase as emissions of halocarbons increased.
Ozone depletion and the ozone hole have generated worldwide concern over increased cancer risks and other negative effects. The ozone layer prevents most harmful UV wavelengths of ultraviolet light (UV light) from passing through the Earth's atmosphere. These wavelengths cause skin cancer, sunburn and cataracts, which were projected to increase dramatically as a result of thinning ozone, as well as harming plants and animals. These concerns led to the adoption of the Montreal Protocol in 1987, which bans the production of CFCs, halons and other ozone-depleting chemicals.
The ban came into effect in 1989. Ozone levels stabilized by the mid-1990s and began to recover in the 2000s. Recovery is projected to continue over the next century, and the ozone hole is expected to reach pre-1980 levels by around 2075.The Montreal Protocol is considered the most successful international environmental agreement to date.
The Connection between Ozone Layer Depletion and UVB Radiation
Reduced ozone levels as a result of ozone depletion ozone depletionA chemical destruction of the stratospheric ozone layer beyond natural reactions. Stratospheric ozone is constantly being created and destroyed through natural cycles. Various ozone-depleting substances (ODS), however, accelerate the destruction processes, resulting in lower than normal ozone levels. The science page (http://www.epa.gov/ozone/science/index.html) offers much more detail on the science of ozone depletion. mean less protection from the sun’s rays and more exposure to UVB
ozone depletionA chemical destruction of the stratospheric ozone layer beyond natural reactions. Stratospheric ozone is constantly being created and destroyed through natural cycles. Various ozone-depleting substances (ODS), however, accelerate the destruction processes, resulting in lower than normal ozone levels. The science page (http://www.epa.gov/ozone/science/index.html) offers much more detail on the science of ozone depletion. mean less protection from the sun’s rays and more exposure to UVB UVBA band of ultraviolet radiation with wavelengths from 280-320 nanometers produced by the Sun. UVB is a kind of ultraviolet light from the sun (and sun lamps) that has several harmful effects. UVB is particularly effective at damaging DNA. It is a cause of melanoma and other types of skin cancer. It has also been linked to damage to some materials, crops, and marine organisms. The ozone layer protects the Earth against most UVB coming from the sun. It is always important to protect oneself against UVB, even in the absence of ozone depletion, by wearing hats, sunglasses, and sunscreen. However, these precautions will become more important as ozone depletion worsens. NASA provides more information on their web site (http://www.nas.nasa.gov/About/Education/Ozone/radiation.html). radiation at the Earth’s surface. Studies have shown that in the Antarctic, the amount of UVB measured at the surface can double during the annual ozone hole.
UVBA band of ultraviolet radiation with wavelengths from 280-320 nanometers produced by the Sun. UVB is a kind of ultraviolet light from the sun (and sun lamps) that has several harmful effects. UVB is particularly effective at damaging DNA. It is a cause of melanoma and other types of skin cancer. It has also been linked to damage to some materials, crops, and marine organisms. The ozone layer protects the Earth against most UVB coming from the sun. It is always important to protect oneself against UVB, even in the absence of ozone depletion, by wearing hats, sunglasses, and sunscreen. However, these precautions will become more important as ozone depletion worsens. NASA provides more information on their web site (http://www.nas.nasa.gov/About/Education/Ozone/radiation.html). radiation at the Earth’s surface. Studies have shown that in the Antarctic, the amount of UVB measured at the surface can double during the annual ozone hole.
 |
Effects on Human Health
Ozone layer depletion increases the amount of UVB that reaches the Earth’s surface. Laboratory and epidemiological studies demonstrate that UVB causes non-melanoma skin cancer and plays a major role in malignant melanoma development. In addition, UVB has been linked to the development of cataracts, a clouding of the eye’s lens.
 Because all sunlight contains some UVB, even with normal stratospheric ozone levels, it is always important to protect your skin and eyes from the sun. See a more detailed explanation of health effects linked to UVB exposure.
EPA uses the Atmospheric and Health Effects Framework model to estimate the health benefits of stronger ozone layer protection under the Montreal Protocol. Updated information on the benefits of EPA’s efforts to address ozone layer depletion is available in a 2015 report, Updating Ozone Calculations and Emissions Profiles for Use in the Atmospheric and Health Effects Framework Model.
Because all sunlight contains some UVB, even with normal stratospheric ozone levels, it is always important to protect your skin and eyes from the sun. See a more detailed explanation of health effects linked to UVB exposure.
EPA uses the Atmospheric and Health Effects Framework model to estimate the health benefits of stronger ozone layer protection under the Montreal Protocol. Updated information on the benefits of EPA’s efforts to address ozone layer depletion is available in a 2015 report, Updating Ozone Calculations and Emissions Profiles for Use in the Atmospheric and Health Effects Framework Model.

Because all sunlight contains some UVB, even with normal stratospheric ozone levels, it is always important to protect your skin and eyes from the sun. See a more detailed explanation of health effects linked to UVB exposure.
EPA uses the Atmospheric and Health Effects Framework model to estimate the health benefits of stronger ozone layer protection under the Montreal Protocol. Updated information on the benefits of EPA’s efforts to address ozone layer depletion is available in a 2015 report, Updating Ozone Calculations and Emissions Profiles for Use in the Atmospheric and Health Effects Framework Model.